As natural elements in aquatic ecosystems, nitrogen and phosphorus support the growth of algae and aquatic plants, providing food and shelter for fish and other organisms. However, when excessive amounts of nitrogen and phosphorus enter water sources, often from human-driven causes, the water quality and surrounding environment can be negatively impacted.
Nutrient pollution in streams, rivers, lakes, and coastal waterways adversely affects aquatic ecosystems and promotes the uncontrolled growth of algal blooms. Algal overgrowth not only blocks light, it consumes dissolved oxygen vital for aquatic wildlife as well. Algal blooms can also release toxins into drinking water supplies, resulting in health issues in human populations.
Approximately 50% of streams in the U.S. contain medium to high levels of nitrogen and phosphorus based on goal background levels of 1 mg/L of nitrogen and 0.1 mg/L of phosphorus, while an estimated 80% of U.S. coastal waterways experience eutrophication, where an excess of nutrients results in plant and algal blooms that deplete oxygen in the water supply. These excessive nutrients are primarily caused by agriculture and industry, including fertilizer runoff, animal manure, sewage treatment plant discharges, storm water runoff, and the combustion of fossil fuels.
Monitoring and assessment of nitrogen and phosphorus concentrations in waterways has become a significant focus for environmental agencies such as the U.S. EPA, which is investigating ways to improve the management and prevention of nutrient pollution. Recently developed in conjunction with the American Society for Testing and Materials (ASTM), “D8001 Test Method for Determination of Total Nitrogen, Total Kjeldahl Nitrogen by Calculation, and Total Phosphorus in Water and Waste Water by Ion Chromatography,” has been approved as a single lab-validated method. This new method allows water quality researchers to concurrently monitor total nitrogen (organic nitrogen, ammonia, nitrate, and nitrite) as nitrate, and total phosphorus as orthophosphate in unfiltered water samples. The method saves time and resources by using a single sample digestion for the simultaneous determination of total nitrogen and total phosphorous.
Separate nitrogen and phosphorus detection
Nitrogen exists as both inorganic and organic species in water. Inorganic nitrogen can be present in its oxidized form as nitrite and nitrate, or in its reduced forms as ammonia and di-nitrogen gas. Organic nitrogen is present in several different sources such as amino acids, proteins, humic acids, and urea. The total nitrogen measurement is the sum of all forms of nitrogen in a sample.
Phosphorus also enters waterways in several forms, including as inorganic orthophosphate, polyphosphate, organic phosphate, and particulate phosphorus. All forms, both organic and inorganic, make up the total phosphorus found in water. Current nutrient detection methods are timeconsuming and costly due to the use of separate procedures for the detection of nitrogen and phosphorus.
Detecting these elements is commonly performed by colorimetric methods based on Kjeldahl digestion, which converts organic nitrogen to ammonia. Determination of the total nitrogen found in a water sample, for example, is the sum of the total Kjeldahl nitrogen (TKN) plus nitrite and nitrate. The TKN method has several well-known issues: environmental safety concerns regarding the use of toxic reagents such as mercury to accelerate decomposition, and negative bias issues if a water sample contains high levels of nitrate. Thus, to determine total nitrogen, two methods are required: one for the determination of TKN, and another for nitrate and nitrite.
There are also several methods for the determination of total phosphorous. Like total nitrogen, these require two steps, digestion and analysis. The digestion step is less complicated than TKN, and a different colorimetric reagent is used for analysis.
Simultaneous nitrogen and phosphorus detection using ion chromatography
To resolve the issues associated with colorimetric methods, ion chromatography (IC) has been adapted to provide an environmentally safer approach that provides the simultaneous detection of both nitrogen and phosphorus. This new ASTM standard uses the powerful separation capabilities of IC combined with improved digestion and filtration techniques for a more sensitive and simplified assay.
IC is a well-established technique for determining inorganic anions in environmental samples. By introducing alkaline persulfate digestion of the sample followed by filtration prior to IC using a high-capacity anion-exchange column, the new method provides safer, simpler, and accurate detection of nitrogen and phosphorus.
Performance of the approach was initially examined by determining recoveries of nitrogen and phosphorus in individual nitrogen- and phosphorus-containing compounds spiked into deionized water. The spike recoveries of these compounds are shown in Tables 1 and 2. Nitrogen recovery ranged from 93 to 100%, indicating good conversion of ammonia and nitrite to nitrate during the alkaline persulfate digestion. Phosphorus recoveries were slightly lower, ranging between 85 and 99%.
Table 1 – Spike recoveries of nitrogen
Table 2 –Spike recoveries of phosphorus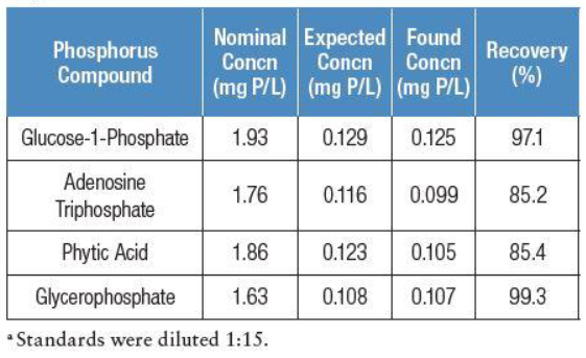
The lower recovery for phosphorus could be attributed to lower purity of test compounds or stability during digestion. These compounds can be used to demonstrate that the digestion is working properly and is complete when spiked into environmental water samples. This is important because the organic N and P amounts in these samples are unknown and it is otherwise difficult to determine if oxidation is complete.
Wastewater sample analysis
Wastewater treatment facilities in the U.S. process an estimated 34 billion gallons of wastewater per day. The water is treated per state and federal regulations and is then released into local waterways, where it can be a source of nutrient pollution. To evaluate the efficacy of combined alkaline persulfate digestion and IC, six different wastewater samples were collected and analyzed from two wastewater facilities.
First the determination of inorganic N and P, as nitrate and phosphate, respectively, was determined by injecting samples into a Thermo Scientific Dionex ICS-2100 Reagent-Free Ion Chromatography System (Thermo Fisher Scientific, Sunnyvale, CA). Second, the samples were digested and analyzed in a similar manner.
The alkaline persulfate digestion procedure, adopted from the United States Geological Survey (USGS), uses equimolar concentrations of persulfate and hydroxide ions to yield samples with a pH >12 after a 1:2 dilution. With initial high-alkaline conditions, nitrogen is oxidized to nitrate. Continued digestion at high temperatures activates bisulfate ions from the decomposition of persulfate to neutralize and then acidify the reaction. With final acidic conditions of pH = 2, any dissolved phosphorus hydrolyzes to orthophosphate.
Once cooled to room temperature, the samples were diluted accordingly and filtered before being injected onto the Thermo Scientific Dionex IonPac AS19 column.
Bisulfate ions produced from the initial digestion and subsequent thermal decomposition of persulfate result in a high concentration of sulfate that can overload standard anion-exchange columns and affect phosphate retention time and recovery. Sample dilution can sometimes override this effect; however, samples with low levels of nitrogen and phosphorus prohibit sufficient dilutions to minimize sulfate interference. Similarly, wastewater samples commonly contain high levels of chloride that is converted to chlorate during digestion. Both residual chloride and the newly formed chlorate also affect column capacity by occupying ion exchange sites on the column.
Applying the high-capacity Thermo Scientific Dionex IonPac AS19 column provided additional capacity and resolution from potential contaminants, including chloride, chlorate, carbonate, and sulfate, thus minimizing the amount of sample dilution and enabling better detection limits. This column separated nitrate and phosphate in less than 20 minutes in the presence of interfering analytes, even with low target element levels.
Nitrite was not detected in any digested samples, most likely because nitrite was converted to nitrate. Table 3 summarizes the wastewater sample results analyzed by IC before and after alkaline persulfate digestion. The nitrate concentration in the undigested samples ranged from 0.5 to 15 mg/L, while the digested samples saw a significant increase in concentration, ranging from 8.5 to 40 mg/L. The difference in phosphate was less dramatic, with the raw water showing the largest increase after digestion. Figure 1 compares the anion separation of the undigested and digested wastewater samples by IC. Note the large increase in the nitrate peak (1) in the digested sample, indicating high amounts of either organic N and/or ammonia.
Table 3 – Wastewater sample results analyzed by IC before and after alkaline persulfate digestion 
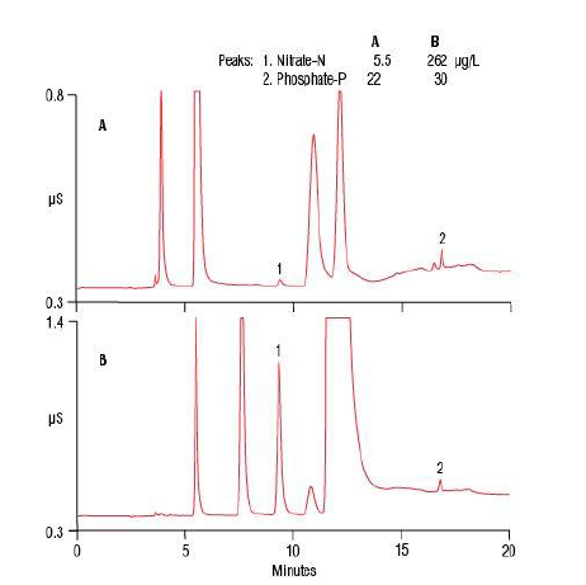 Figure 1 – Separation of anions in an undigested (A) and alkaline persulfate digested (B) raw wastewater sample by ion chromatography
Figure 1 – Separation of anions in an undigested (A) and alkaline persulfate digested (B) raw wastewater sample by ion chromatographyMethod linearity, limits of detection, and limits of quantification were determined to establish the lowest concentration that can be measured reliably using this approach. Using the Thermo Scientific Dionex EGC III KOH eluent generator cartridge as part of the IC system provided suppressed background conductivity of 0.35 µS at the start of analysis with a very slight increase to 0.39 µS by the end of analysis. Low background conductivity generates a lower baseline noise that enables low detection limits of 0.76–1 µg/L for nitrogen and 1.3 µg/L for phosphorus.
Conclusion
Detection of total nitrogen and total phosphorus has been demonstrated and led to the development of the new ASTM standard. Supplementing alkaline persulfate digestion and ion chromatography with a high-capacity anion-exchange column and a hydroxide eluent using electrolytic eluent generation and suppression creates a unique combination of tools that allow for lower detection limits and increased sensitivity. The method resolves environmental and safety issues associated with previous methods, and simplifies detection protocols by combining nitrogen and phosphorus analysis, while contributing to the management and prevention of nutrient pollution.
Additional reading
- https://www.astm.org/Standards/D8001.htm
- https://www.epa.gov/sites/production/files/2015-08/documents/method_365-1_1993.pdf
- https://www.epa.gov/sites/production/files/2015-06/documents/epa-350.1.pdf
- https://www.epa.gov/sites/production/files/2015-10/documents/method_1687_draft_2001.pdf
- https://archive.epa.gov/water/archive/web/html/vms56.html
Jeff Rohrer and Richard Jack are with Thermo Fisher Scientific, 1214 Oakmead Pkwy., Sunnyvale, CA, U.S.A.; www.thermofisherscientific.com. Brian De Borba is with AbbVie, Chicago, IL, U.S.A.